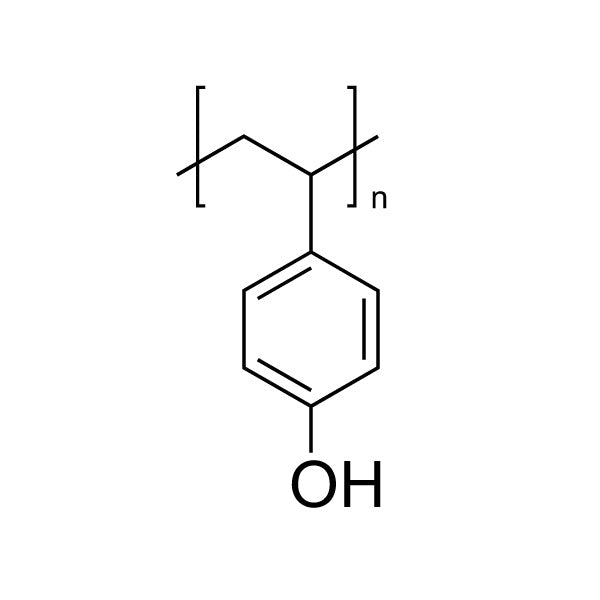
Poly(4-vinylphenol)
Product Number:
CAS #
-
06527
-
24979-70-2
Product Number:
CAS #
-
06527
-
24979-70-2
Product Description
Poly(4-vinylphenol) (PVP)
Poly(4-vinylphenol) (PVP) is a thermoplastic polymer characterized by a high glass transition temperature, strong hydrogen-bonding capability, and adhesion to a wide range of substrates. The presence of phenolic hydroxyl groups along the polymer backbone contributes to its polarity, intermolecular interactions, and surface activity.
Due to these properties, poly(4-vinylphenol) is commonly used as a functional resin in coatings, adhesives, and photoresist systems. Its film-forming behavior, chemical functionality, and thermal characteristics make it a widely studied material in electronic materials and surface-modification research.
Key Properties
- Polymer type: Poly(4-vinylphenol)
- Polymer class: Thermoplastic aromatic polymer
- Functional groups: Phenolic hydroxyl groups
- Glass transition temperature: High (relative to many vinyl polymers)
- Intermolecular interactions: Strong hydrogen bonding
- Surface behavior: Adhesive to a variety of substrates
- Typical use: Research and formulation development
Applications
-
Photoresists and electronic materials
Studied for use in semiconductor processing, microfabrication, and electronic material systems where thermal stability and functional surface chemistry are required -
Adhesives and surface modifiers
Evaluated as a functional resin to enhance adhesion strength in coatings, composites, and interfacial layers -
Biomedical and hydrophilic coatings
Investigated for use in biomaterials and surface coatings where increased wettability and polar surface interactions are desired
FAQs
Common questions about Poly(4-vinylphenol).
-
What is poly(4-vinylphenol) used for?
Poly(4-vinylphenol) is commonly studied as a functional polymer in coatings, adhesives, and photoresist systems, particularly where thermal stability, hydrogen bonding, and surface adhesion are important.
-
Why does poly(4-vinylphenol) exhibit strong adhesion?
The phenolic hydroxyl groups along the polymer backbone enable hydrogen bonding and polar interactions with many substrates, contributing to adhesion and surface activity.
-
What role does poly(4-vinylphenol) play in photoresist formulations?
In photoresist research, poly(4-vinylphenol) is studied for its film-forming properties, thermal behavior, and chemical functionality, which can influence pattern formation and processing performance.
-
Is poly(4-vinylphenol) hydrophilic?
The phenolic hydroxyl groups impart polarity and hydrophilic character relative to non-functional vinyl polymers, although overall wettability depends on formulation and surface conditions.
-
Why is poly(4-vinylphenol) considered a high-Tg polymer?
The rigid aromatic backbone and strong intermolecular interactions restrict chain mobility, resulting in a higher glass transition temperature compared to many aliphatic vinyl polymers.